Work Order Management
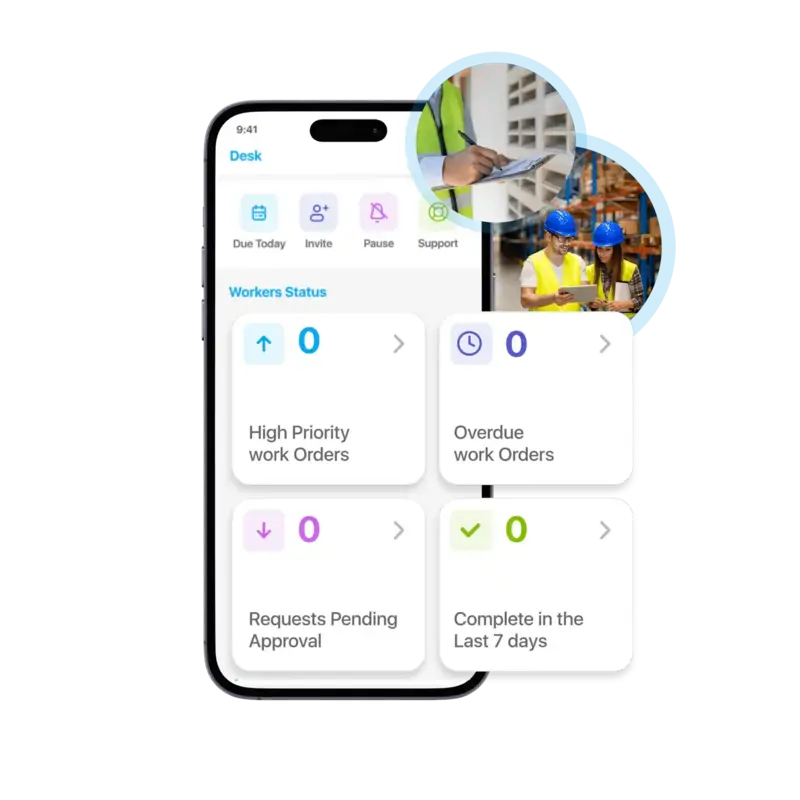
Create, assign, prioritize, and track every work order from a single dashboard. Auto-assign tasks based on technician availability and skill set to maximize efficiency.
Task360 is a state-of-the-art Computerized Maintenance Management System built for modern teams. Streamline work orders, manage assets, schedule preventive maintenance, and leverage AI-powered insights, all from one platform.
Organizations relying on spreadsheets and manual processes are bleeding time and money without realizing it. Outdated systems create compounding problems that affect every layer of operations.

Task360 offers an intuitive platform for creation, assignment, and tracking of every work order from a single unified dashboard. Get real-time insights at your fingertips and empower your field teams with a mobile-first experience designed for how maintenance actually happens.

Nine powerful modules built to cover every aspect of maintenance management, from first request to final report.
Create, assign, prioritize, and track every work order from a single dashboard. Auto-assign tasks based on technician availability and skill set to maximize efficiency.
Maintain complete lifecycle records for every asset including maintenance history, usage stats, technical specs, and warranty info. Never lose visibility of critical equipment.
Eliminate reactive maintenance with automated PM schedules triggered by time, meter readings, or condition thresholds. Catch problems before they become costly failures.
Task360's AI analyzes historical data to predict failures before they happen and turn anomalies, notes, and inspection findings into ready-to-review maintenance tasks. Let machine learning help your team act faster on critical assets.
Track spare parts levels, set automatic reorder points, and link inventory directly to work orders. Ensure your technicians always have the right parts on hand.
Make data-driven decisions with 40+ pre-built reports covering KPIs, technician performance, asset health, and maintenance costs. Turn maintenance data into actionable insight.
Getting started with Task360 is fast and straightforward. No lengthy implementations, no consulting fees.
Create your account, import your assets and locations, and configure your team structure. Our onboarding wizard guides you through every step.
Define your work order templates, PM schedules, inspection checklists, and notification rules. Use our pre-built templates or create your own.
Your team starts using Task360 immediately on web and mobile. Track KPIs from day one and continuously optimize with AI-powered insights.
Not sure if you're ready? Take our free readiness assessment →
Purpose-built modules that work together seamlessly to cover every dimension of maintenance management.
The core of Task360, manage every work order, PM schedule, and maintenance request from a single, intuitive dashboard designed for maintenance teams of all sizes.
Learn more →Track the full lifecycle of every asset from procurement to retirement. Monitor health, depreciation, warranties, and maintenance history in one centralized place.
Learn more →Dispatch field technicians, track job status in real time, capture photos and signatures on-site, and sync everything back automatically in real time.
Learn more →Build custom inspection checklists, schedule recurring rounds, capture findings with photos, and generate compliance-ready reports instantly.
Learn more →With Task360's mobile-first design, your technicians can create work orders, update asset records, and complete inspections directly from their smartphone, with real-time sync across all devices.
Task360 adapts to the unique maintenance workflows and compliance requirements of your sector.
Everything you need to know about Task360 and how it can work for your organization.
Task360 is an advanced maintenance management system designed to streamline and optimize maintenance operations. It enhances asset longevity, reduces downtime, and ensures regulatory compliance through a comprehensive, all-in-one solution.
Task360's predictive maintenance tools and real-time monitoring help identify issues before they become failures, significantly reducing unplanned downtime across all your assets and facilities.
Customers typically report a 20-30% reduction in maintenance costs, a 24%+ reduction in equipment downtime, and significant improvements in team productivity within the first 90 days of using Task360.
Yes. Task360 offers API access and extensive integration options including ERP systems, IoT sensors, and business management platforms, ensuring a smooth fit into your existing technology stack.
Task360 provides dedicated account managers, 24/7 technical support, comprehensive onboarding assistance, and ongoing training resources to ensure your team gets the most from the platform.
Join countless organizations choosing Task360 for modern, efficient, and intelligent maintenance management. Schedule your demo today.
Book a Demo